Mountain pine beetle (factsheet)
We have archived this page and will not be updating it.
You can use it for research or reference.

French common name: Dendroctone du pin ponderosa
Scientific name: Dendroctonus ponderosae Hopkins
Order: Coleoptera
Family: Curculionidae
Distribution
The mountain pine beetle is native to western North America, from northern Mexico to northern British Columbia. It is also present in an isolated population of pine that is surrounded by prairie in the Cypress Hills area of southwestern Saskatchewan, which was likely infested in the early 1980s. In the 2000s, the beetle significantly expanded its range in Canada, invading new habitat east of the Rocky Mountains in northeastern British Columbia and northern Alberta.
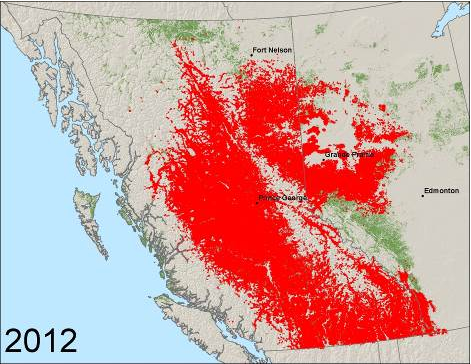
Observed presence of mountain pine beetle from 1999 to 2012. Map data: Forest Practices Branch, Ministry of Forests and Range, Government of British Columbia; Environment and Sustainable Resource Development, Government of Alberta; Forest Insects and Disease Survey, Pacific Forestry Centre, Canadian Forest Service, Natural Resources Canada.
Hosts in Canada
Principal hosts in Canada
Lodgepole pine is the most common host in the mountain pine beetle’s range in British Columbia.
Other hosts
Most species of pine that grow in the beetle’s range are readily attacked, with the exception of Jeffrey pine. In Canada, these hosts include ponderosa, western white, whitebark and limber pines and, very recently, jack pine in the expanded range.
Occasional or potential hosts
Most pine species native to Canada, as well as four non-native species, have been successfully attacked when planted in the beetle’s range in western North America. Non-pine hosts, such as interior spruce, Douglas-fir and western larch, are sometimes attacked during outbreaks when they grow with pine, but beetle populations do not persist long-term in non-pine hosts.
Life history

The mountain pine beetle has a one-year life cycle in most of its range, but may take more or less time to complete its development, depending on local temperatures. Adult beetles usually disperse in July or August, depending on the region, to colonize new host trees. Females attack first and release semiochemicals called aggregation pheromones that attract more females and males to the tree. Female beetles lay eggs along the sides of vertical galleries that they excavate in the inner bark of the tree. Newly hatched larvae mine away from the egg galleries. Insects usually overwinter as larvae, completing their development the following spring and pupating in June or July.
During gallery construction, fungal symbionts carried by beetles in specialized pockets in one of the mouthparts are introduced to the trees. The fungi colonize the inner bark and sapwood, interrupting tree function and defence in addition to changing the moisture and chemistry of tree tissues in which insects are developing. The fungi sporulate in pupal chambers and new adults feed on the spores before emerging and dispersing to a new host tree.
Ecology
Trees defend themselves against mountain pine beetle attack with toxic resin. Low or endemic beetle populations cannot overcome the defences of healthy trees and attack suppressed, weak or dying trees. Suppressed and weak trees are usually poor-quality hosts for the beetles because they may already have been attacked by competing insects and the thin inner bark layer is a poor habitat.

As populations increase, the mountain pine beetle is able to overwhelm the defences of larger and healthier trees through a rapid, coordinated group or mass attack. Large trees provide better habitat and produce more beetles. This results in positive feedback and rapid population growth. Tree defences may be important in regulating low or endemic populations, but they become inconsequential when beetle populations are high. Landscape-level epidemics only decline once most of the large diameter host trees have been killed or unfavourable weather causes catastrophic insect mortality.
In British Columbia, the northern limit of the beetle’s range has been limited by cold winter temperatures (−40°C) and cool summers. Outbreaks have been linked to favourable weather in both summer and winter. Warm, dry summers are good for beetle development and dispersal, and drought stress reduces tree defences. Overwinter mortality is usually the largest single source of mountain pine beetle mortality; mild winter temperatures result in higher insect survival. Successive years of favourable summer and winter weather combined with an abundance of mature suitable pine hosts on the landscape have been cited as factors contributing to the massive epidemic that occurred in the 1990s and 2000s in British Columbia.
Attack and damage
Sawdust on the outer bark around beetle entrance holes is the first sign of attack. Pitch tubes (small globs of pitch) are present on the outer bark of attacked trees within days of attack and remain visible for many years; however, pitch tubes may not be present on trees with severely compromised defences. The sapwood of successfully attacked trees appears blue in colour usually by the fall in the year of attack. Needles turn yellow, orange and then red approximately one year after attack. The crowns of some trees may start to fade in the same year as the attack, depending on environmental conditions. The red needles drop off approximately two to four years after attack and trees appear gray with no needles.

Status in Canada
The largest recorded mountain pine beetle epidemic occurred in the 1990s and 2000s in British Columbia. Over 18 million hectares of forest were impacted to some degree, resulting in a loss of approximately 723 million cubic metres (53%) of the merchantable pine volume by 2012. The epidemic peaked in 2005: total cumulative losses from the outbreak are projected to be 752 million cubic metres (58%) of the merchantable pine volume by 2017, when the epidemic will have largely subsided in British Columbia.
Several times in the 2000s, beetles from the massive epidemic in central British Columbia were carried on upper atmospheric winds across the biogeoclimatic barrier posed by the Rocky Mountains. The insects were deposited in northeastern British Columbia and northwestern Alberta. These long-distance dispersal events resulted in a significant increase in the distribution of the mountain pine beetle in Canada. The beetle is now established in lodgepole–jack pine forests in northern Alberta and threatens to spread east across Canada’s boreal forest if conditions are favourable. The mountain pine beetle has also moved northwards and in 2012 was reported north of 60° latitude in the Northwest Territories for the first time, although the fate of this small population is uncertain.

Contact: Kathy Bleiker
Links
Canadian Forest Service publications on mountain pine beetle
References
References
Cerezke, H.F., and F.J. Edmond. 1989. Forest insect and disease conditions in Alberta, Saskatchewan, Manitoba, and the Northwest Territories in 1987. Information Report NOR-X-300. Edmonton, Alberta: Northern Forestry Centre, Canadian Forest Service, Natural Resources Canada.
Furniss, M.M., and J.A. Schenk. 1969. Sustained natural infestations by the mountain pine beetle in seven new Pinus and Picea hosts. Journal of Economic Entomology 62: 518–519.
Paine, T.D., K.F. Raffa, and T.C. Harrington. 1997. Interactions among scolytid bark beetles, their associated fungi, and live host conifers. Annual Review of Entomology 42: 179–206.
Raffa, K.F., B.H. Aukema, B.J. Bentz, A.L. Carroll, J.A. Hicke, M.G. Turner, and W.H. Romme. 2008. Cross-scale drivers of natural disturbances prone to anthropogenic amplification: the dynamics of bark beetle eruptions. BioScience 58: 501–517.
Safranyik, L., and A.L. Carroll. 2006. The biology and epidemiology of the mountain pine beetle in lodgepole pine forests. In L. Safranyik and B. Wilson (eds.), The mountain pine beetle: A synthesis of its biology, management and impacts on lodgepole pine. Victoria, BC: Pacific Forestry Centre, Canadian Forest Service, Natural Resources Canada.
Safranyik, L., A.L. Carroll, J. Régnière, D.W. Langor, W.G. Riel, B. Peter, B.J. Cooke, V.G. Nealis, S.W. and Taylor. 2010. Potential for range expansion of mountain pine beetle into the boreal forest of North America. Canadian Entomologist 142: 415–442. doi:10.4039/n08-CPA01.
Smith, R.H., J.P. Cramer, and E.J. Carpender. 1981. New record of introduced hosts for the mountain pine beetle in California. Research Note PSW-354, United States Forest Service.
Walton, A. 2013. Provincial-level projection of the current mountain pine beetle outbreak: update of the infestation projection based on the Provincial Aerial Overview Surveys of Forest Health conducted from 1999 through 2012 and the BCMPB model (year 10). Victoria, BC: BC Ministry of Forests, Lands and Natural Resources Operations.
Wood, S.L. (ed.). 1982. The bark and ambrosia beetles of North and Central America (Coleoptera: Scolytidae): a taxonomic monograph. Provo, Utah: Brigham Young University.
Page details
- Date modified: